Dr. Bae served as a principal researcher for coflex’s fda approval
Did you know…
Dr. Bae was the principal investigator for coflex®, the first and only motion preserving and minimally invasive treatment approved by the FDA for moderate to severe spinal stenosis after decompression?
The Authority on coflex® Interlaminar Stablization
Named “spine surgeon leader to know” by Becker’s Spine Review, Dr. Hyun Bae is leading the shift from spinal fusion to motion-preserving technologies such as coflex®. While physicians throughout the nation have received training on the coflex® interlaminar technology, Dr. Bae has been involved with the device since infancy. He was among a prestigious group of surgeons selected to develop and implement coflex® and served as principal researcher during clinical trials for its FDA approval. As the author of numerous peer review articles on coflex®, Dr. Bae shares the latest in spinal surgery with scientists, surgeons and health care professionals across the globe.
“Dr. Bae has a way about him – calming, reassuring, on the cutting edge of things. I can tell you that leading up to the surgery, the surgery and the follow-up care afterward was pretty amazing. I’ve got no complaints.”
(– Ken Josefsberg Ironman competitor, runner and avid golfer)
What Should I Expect After Lumbar Surgery?
Coflex® is a flexible, u-shaped titanium alloy device designed to restore mobility and stop back pain resulting from spinal stenosis. During decompression, the FDA approved device is inserted between two vertebrae, relieving pressure and restoring the narrowed space caused by a compressed nerve or collapsed disc. Because coflex® acts as a shock absorber, the device supports the natural movement of the spine. Unlike traditional fusion, there are no screws to immobilize or fuse the back, thus maximizing range of motion and promoting a faster recovery period.
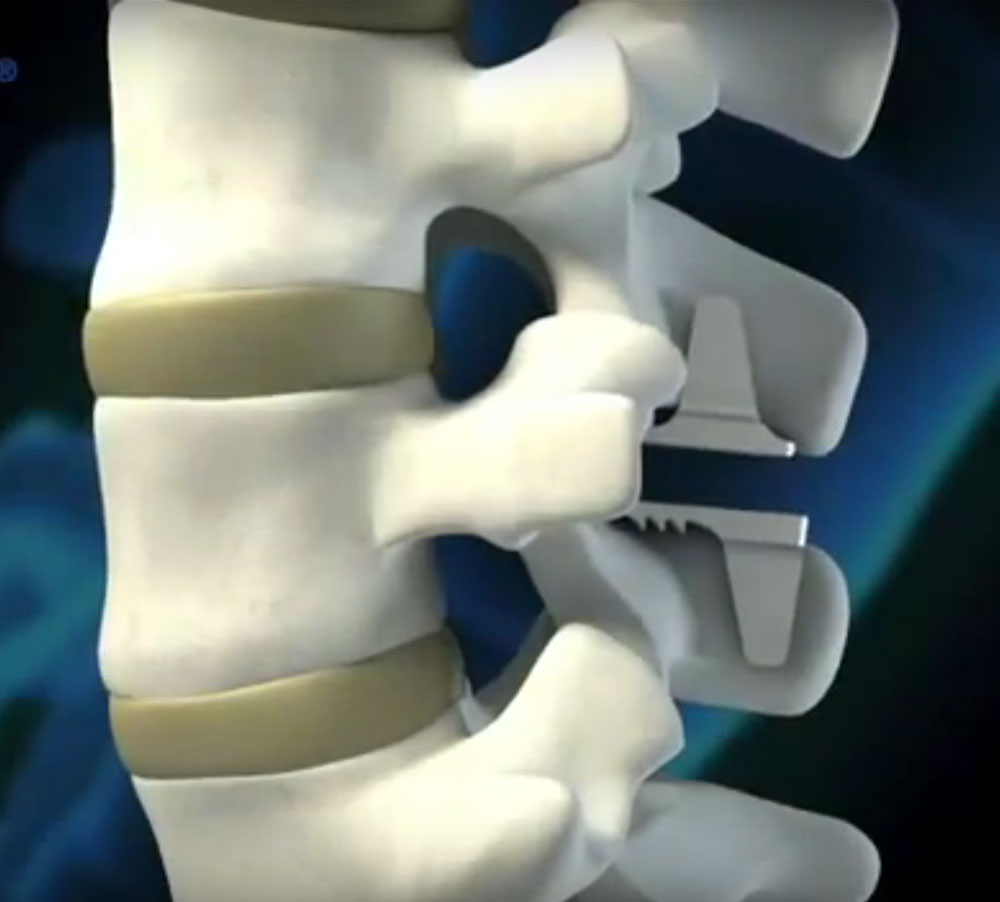
Is Decompression with Coflex Effective?
“As a clinician researcher, I want to bring my research to the patients as soon as possible.” Dr. Bae has achieved this goal, participating in dozens of clinical trials proving the effectiveness of coflex® for treating low-grade spondyloisthesis and lumbar spinal stenosis. Two-year clinical and radiographic results from the US IDE trial showed that coflex® had a statistically superior outcome over those patients treated with fusion at two years’ post-operation. Surgery was also less complex and invasive, had a lower rate of complications, a shorter length of hospital stay and a faster recovery period. Adjacent segment stress was also significantly reduced with coflex®.
Are You a Candidate for the coflex® Interlaminar Technology?
Orthopedic medicine is evolving quickly and new devices are rapidly taking over traditional spinal fusion surgery. With technology and cutting-edge advancements increasing the options for patients looking to minimize chronic back pain, it’s important to work with a highly qualified board-certified surgeon.
Dr. Bae is the authority on coflex® with extensive knowledge in how the device works as well as the most effective and safe applications. To see if you’re a candidate for the coflex® interlaminar technology, contact The Spine Institute Center for Spinal Restoration at (310) 828-7757.